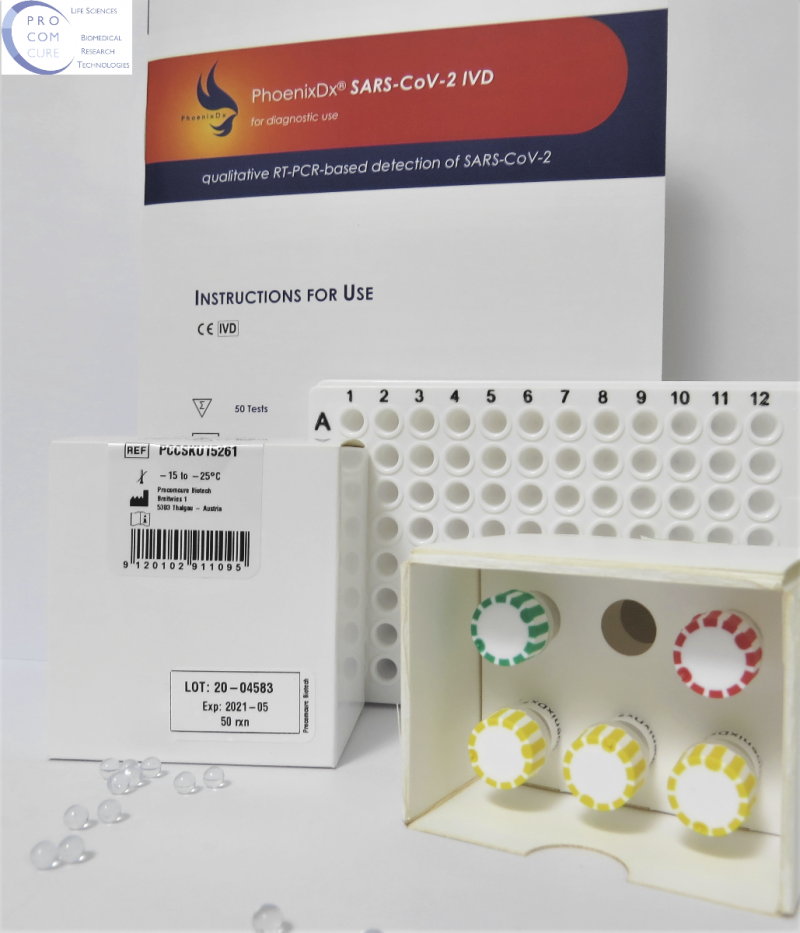
PhoenixDx® SARS-CoV-2 IVD
Auf Lager
SKU/Catalog Number
PCCSKU15261
Katalognummer: PCCSKU15261
Method: RT-PCR
| Intended Use | PHOENIXDX® SARS-COV-2 IVD is a real-time RT-PCR-based diagnostic test for the in vitro qualitative detection of 2019-Novel Coronavirus (SARS-CoV-2) in respiratory specimens and sera from patients who meet COVID-19 clinical and/or epidemiological criteria. |
|---|---|
| Size | 50 Tests / Kit |
| Expiry date | 10-12 Monate |
| Certification | CE IVD |
| Company | Procomcure Biotech GmbH |
| Storage condition | -20°C |
| Materials Provided |
|
| Product Number | PCCSKU15261 |
| Hersteller | Procomcure Biotech GmbH |
| Zertifizierung | CE IVD |
| Order | Request Quote |